Poster sessions

Dopamine 2022
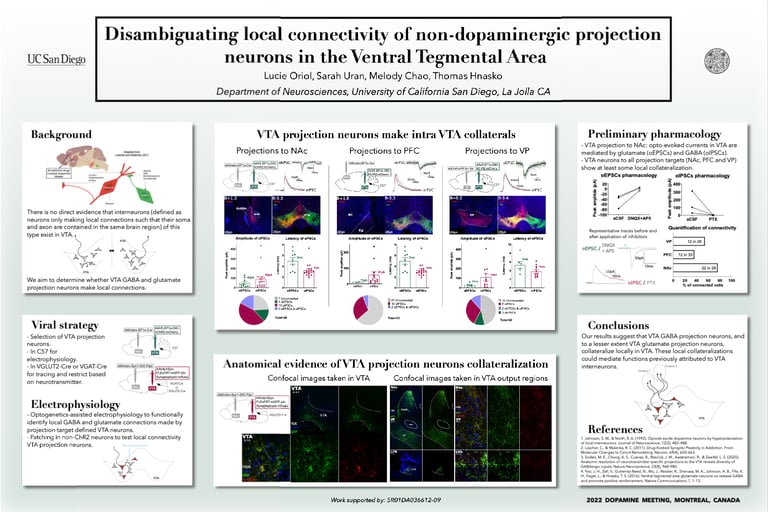
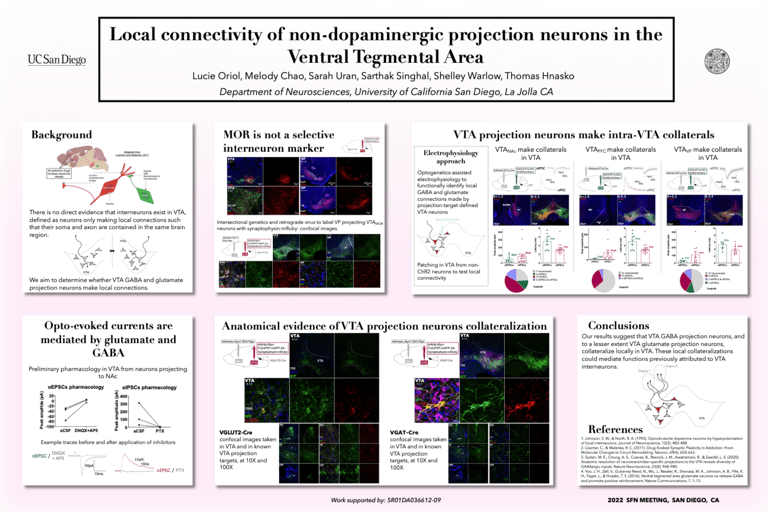
The ventral tegmental area (VTA) is central to brain reward circuits and addictive drugs share a common effect of increasing dopamine released from VTA projections, most famously in nucleus accumbens (NAc). Understanding the VTA functional organization is therefore important toward understanding the mechanisms underlying addiction. For example, opioids reduce GABA-mediated inhibition in the VTA, thereby disinhibiting dopamine neurons, which contributes to the reinforcing properties of those drugs. While classical models suggest that dopamine disinhibition in VTA is mediated by GABA interneurons (defined as neurons only making local connections such that their soma and axon are contained in the same brain region), there is no direct evidence that interneurons of this type exist in VTA. Instead, GABA projection neurons in VTA could make intra-VTA collaterals. To determine whether VTA GABA and glutamate projection neurons make local connections, we used optogenetics-assisted electrophysiology to functionally identify local GABA and glutamate connections made by projection-target defined VTA neurons. We selectively expressed ChR2 in VTA projection neurons (to NAc, prefrontal cortex, and ventral pallidum successively) and performed whole-cell patch clamp recordings of VTA neurons. We detected opto-evoked excitatory postsynaptic currents, presumably mediated by glutamate, and opto-evoked inhibitory postsynaptic currents, presumably mediated by GABA. Our results suggest that VTA projections neurons collateralize to make intra-VTA connections (as well as distal ones) which could mediate functions prior attributed to VTA interneurons.

Society for Neuroscience 2022
The ventral tegmental area (VTA) is central to brain reward circuits and addictive drugs share a common effect of increasing dopamine released from VTA projections, most famously in nucleus accumbens (NAc). Understanding the VTA functional organization is therefore important toward understanding the mechanisms underlying addiction. For example, opioids reduce GABA-mediated inhibition in the VTA, thereby disinhibiting dopamine neurons, which contributes to the reinforcing properties of those drugs. While classical models suggest that dopamine disinhibition in VTA is mediated by GABA interneurons (defined as neurons only making local connections such that their soma and axon are contained in the same brain region), there is no direct evidence that interneurons of this type exist in VTA. Instead, GABA projection neurons in VTA could make intra-VTA collaterals. To determine whether VTA GABA and glutamate projection neurons make local connections, we used optogenetics-assisted electrophysiology to functionally identify local GABA and glutamate connections made by projection-target defined VTA neurons. We selectively expressed ChR2 in VTA projection neurons (to NAc, prefrontal cortex, and ventral pallidum successively) and performed whole-cell patch clamp recordings of VTA neurons. We detected opto-evoked excitatory postsynaptic currents, presumably mediated by glutamate, and opto-evoked inhibitory postsynaptic currents, presumably mediated by GABA. Our results suggest that VTA projections neurons collateralize to make intra-VTA connections (as well as distal ones) which could mediate functions prior attributed to VTA interneurons.

International Narcotics Conference 2025
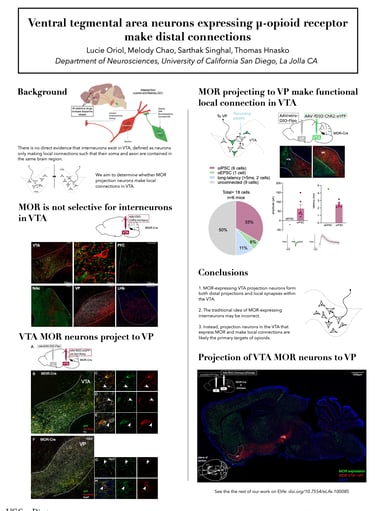
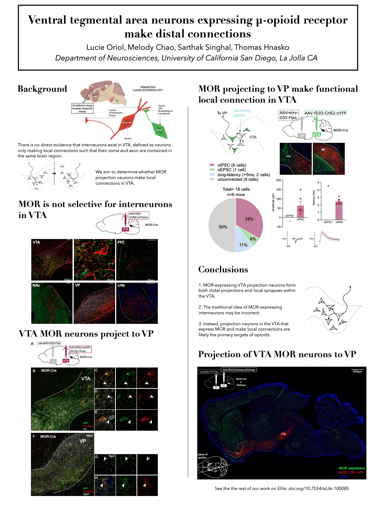
The ventral tegmental area (VTA) is central to brain reward circuits and a major target for opioids and other drugs of abuse. VTA is famous for dopamine neurons, but also contains GABA- and glutamate-releasing neurons, each of which can mediate different behavioral effects depending on their projection targets. Untangling the connectivity of heterogeneous VTA neurons is thus crucial to understanding mechanisms underlying addiction. Opioids and other addictive drugs share a common effect of increasing dopamine released from VTA projections. The µ-opioid receptor (MOR) is the most important target for both opioid analgesia and addiction. Opioids are known to reduce GABA-mediated inhibition in the VTA, thereby disinhibiting dopamine neurons, which contributes to the reinforcing properties of those drugs. While classical models suggest that dopamine disinhibition in VTA is mediated by MOR-expressing inhibitory interneurons, defined as neurons only making local connections such that their soma and axon are contained in the same brain region, there is no direct evidence that such interneurons exist in VTA. Instead, MOR-expressing projection neurons in VTA could make intra-VTA collaterals. To elucidate whether MOR is a selective marker for interneurons, we selectively expressed a membrane-bound fluorescent protein in MOR-expressing VTA neurons, revealing dense projections to ventral pallidum (VP) as well as to other brain regions. To determine whether MOR-expressing projection neurons make local connections, we used optogenetics-assisted electrophysiology to functionally identify local GABA connections made by projection-target defined VTA neurons. We found that VTA neurons expressing MOR make distal connections, and that these projection neurons also make local intra-VTA synapses. Our findings suggest that VTA projection neurons, rather than VTA interneurons, may be a major target for opioids.